“Scalpel, please,” the microsurgeon says as she prepares to make an abdominal incision to extract the heart from the donor mouse. She has no more than four minutes to excise the still-beating heart for transplant into the recipient. Once the successful transplantation is complete, the recipient mouse now possesses a functioning yet foreign organ.
Unlike mice, human transplant patients can face serious lifelong health challenges due to essential medication used to suppress their immune system so they do not reject the unfamiliar transplant. And although some recipients will live for decades with a transplanted organ, some unfortunately reject it many years down the line. Rejection here means that the transplanted organ cells die and thus the organ loses function, often due to immune-mediated events. Scientists like Jen Allocco are working to learn why this seemingly spontaneous transplant rejection occurs.
“There are thousands of people on our national waiting list, so we really can’t afford to have more than one transplantation per person,” said Allocco. “So when a patient gets a transplant, we want them to keep it.”
Your immune system attacks things that aren’t you
To understand why a body might reject an unfamiliar transplanted organ, it is necessary to first understand what normally happens when immune cells encounter something foreign. Ordinarily, immune cells need two signals to know that it is time to attack an invader, a process called immune cell activation.
Signal One is a foreign piece of biological material, that is, something like DNA or protein that is non-self (as you might imagine, the body normally doesn’t attack its own cells). Signal Two is some sort of cellular damage or sign of infection. Allocco, a senior graduate student at the University of Chicago studying transplant immunology in Maria-Luisa Alegre’s lab, explained that in order to mount an immune response, “the body needs Signal Two. There needs to be context that there’s an infection or something bad going on.”
This explains why the body doesn’t mount an immune response to just anything that enters it, like food.
“When you eat something new, there’s no inflammation,” said Allocco. “There’s nothing to tell your immune system that you should attack that thing.” In this case, the immune cells are receiving Signal One but not Signal Two, so there’s no immune response and, as a result, no inflammation.
In the context of a transplant, not only is a foreign biological material being introduced—someone else’s organ or tissue—but the recipient is undergoing damage.
“Basically, you’re cutting someone open. You’re putting something foreign in and there’s damage associated with it, and that’s sufficient for your immune cells to activate,” Allocco said.
How to reject transplant rejections
“Transplants are kind of a paradox because normally you don’t want to put something foreign into your body,” said Allocco. So how do patients who receive transplants survive for years without rejecting the foreign organ or tissue?
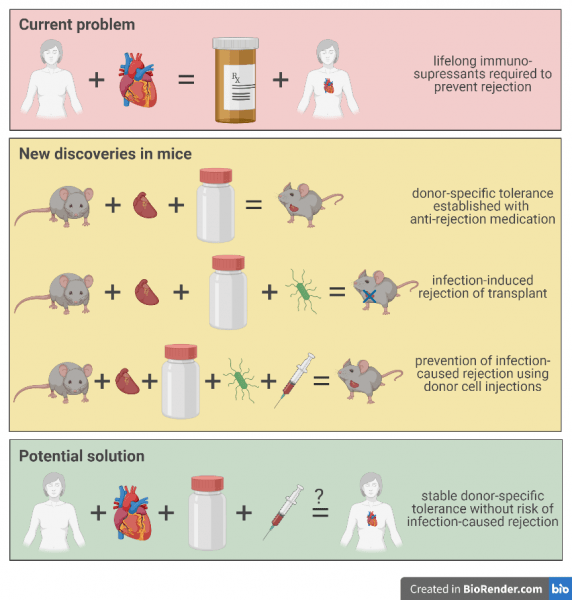
As Allocco explained, “what we have now is lifelong immunosuppression,” meaning transplant recipients take medication to ramp down their immune system. This allows patients to survive for years with their new organs or grafts, but it also puts them at risk of infection. That’s because the same functions that are suppressed to prevent transplant rejection are also responsible for fighting bacterial and viral infections. As a result, transplant survivors are less likely to be able to fight off and survive infections than people who are not on immunosuppressive medications.
Mice do not require immunosuppression to prevent transplant rejection
Because of this risk, Allocco and her colleagues are motivated to understand the interplay between transplants, infections, and medications that prevent organ rejection. To study this interplay, the Alegre lab uses a model system in which transplant recipients do not require lifelong immunosuppression. Interestingly, mice, the model organisms, can avoid long-term immunosuppression medication after a transplant or graft. Instead, mice can establish something called donor-specific tolerance with the use of a set of anti-rejection medications taken over a much shorter time period than that of typical immunosuppressants prescribed to humans.
Allocco explained that when their immune system is non-responsive, mice can achieve donor-specific tolerance to only the individual who gave the graft. In other words, by using a set of anti-rejection drugs for a short time, the recipient’s immune system won’t activate a response to the donor’s cells specifically, but will still continue to operate as a normal immune system otherwise, such as in the case of an infection.
If scientists and doctors could use the same anti-rejection medications in humans as they do in mice to treat transplant recipients, then they may be able to establish donor-specific tolerance in humans, and perhaps these patients could live longer and with higher quality of life.
If mice can keep their transplants, why can’t we?
The bad news is that the specific set of anti-rejection medications given to establish donor-specific tolerance in mice doesn’t work in humans.
While scientists work to improve the medication to establish donor-specific tolerance in humans, patients must remain on a strict regimen of immunosuppressants for their entire life, putting them at high risk of suffering or even dying from an infection.
At the same time, the Alegre lab came across a puzzling phenomenon in a small group of kidney transplant survivors in France who had actually stopped taking their immunosuppressant medication. Amazingly, these patients spontaneously developed donor-specific tolerance of their transplants, similar to what Allocco observes in her mice.
However, after years of living with donor-specific tolerance, some of the recipients’ immune systems suddenly rejected their transplanted kidney years later. Interestingly, this rejection was correlated with those patients who had experienced a serious infection.
It is remarkable that human transplant recipients were able to develop donor-specific tolerance, but concerning that infections were correlated with sudden rejection. “If you think about it, it should be a permanent shutoff of the immune response to the transplant,” Allocco said.
As scientists and doctors get closer to being able to establish donor-specific tolerance in humans with new drug development, the post-tolerance rejection represents an urgent issue. Given the ever-growing organ waitlists in the United States, doctors and patients do not need the additional burden of a high rate of transplant rejections. On top of that, it is very emotionally and physically stressful for patients to undergo a major surgery only to reject their transplant and then have to return to an already full waitlist. “It’s really tragic,” said Allocco.
But how might scientists study the interplay between infections and transplants in humans? “Obviously we’re not going to be giving humans infections to see if their graft rejects,” Allocco joked. Instead, she wondered if she could replicate this phenomenon of infection-induced rejection in mice who already have established donor-specific tolerance.
The connection between infections and transplant rejections
To figure out if previously established tolerance disappears, Allocco takes mice that have achieved donor-specific tolerance of heart transplants and infects them with bacteria to test whether or not the recipients suddenly begin to reject the donor’s heart. Indeed, the infected mice very often reject their transplants when compared to uninfected mice.
Allocco’s colleagues in the Alegre lab have shown that during donor-specific tolerance, the immune cells that specifically target the transplant become dysfunctional when confronting the foreign tissue and thus do not mount an immune response against it. This is what leads to tolerance of the transplant rather than immune-mediated rejection.
After an infection, Allocco and her colleagues hypothesize that those dysfunctional immune cells actually suddenly become semi-functional against the transplant, which may lead to rejection.
However, the Alegre lab also discovered that exposing the recipient mice to their donor’s immune cells for several weeks after the transplant could actually prevent the transplant rejection observed post-infection. In other words, after tolerance was established, donor mouse cell injections were protective from the rejection that was ordinarily observed after an infection.
This result presents an exciting opportunity to prevent rejection in human transplant patients: perhaps repeated donor blood transfusions could improve tolerance.
Currently, people need life-long regimens of immunosupressants after an organ transplant. New discoveries in mice models suggest that donor-specific tolerance can be established.
Innovative research can positively impact patient lives in the short-term
Understanding how and why infection-induced rejection happens in mice will help inform therapeutic strategies to prevent rejection in humans who have developed donor-specific tolerance.
“We’re one of the only ones who are looking at this semi-rejection where you have tolerance and then you lose tolerance,” Allocco said. She hopes one day all recipients will achieve donor-specific tolerance towards their transplants, making them less likely to reject their new tissue later on in life. Allocco explained that her lab’s research is focused on asking how they can make transplants persist in patients for longer. To answer this, they are studying the different aspects of tolerance which will likely someday be translated for the clinic.
The motivation for this research comes easily to scientists
Allocco likes the fact that the time from the lab bench to patient bedside is relatively short. “I really like that it’s very translational research,” she said.
Allocco also finds the work intellectually engaging because, as she explained, “we are able to ask really interesting questions using our model. I can put my academic learning to use, and I like that. I just really like science.”
Besides enjoying the science, Allocco knows several people who have received successful transplants, including a heart transplant. Seeing first-hand the difference these successes make in people’s lives further motivates Allocco to devote herself to her research. Allocco’s work may one day lead to low risk of transplant rejection, meaning she and others can rest easy concerning the state of their transplants.
Due to curious people like Allocco who are excited by what they do, there may soon be improvements in the long-term outcomes and quality of life of transplant patients.
By Sammy Keyport
Sammy Keyport is a PhD candidate at the University of Chicago in the Genetics, Genomics, and Systems Biology program. Sammy works in Dr. D. Allan Drummond’s lab studying how cells sense stress.
This article was written in collaboration with the myCHOICE Writing for Communications and Marketing Careers workshop, hosted by Marina Damiano of the Damiano group.