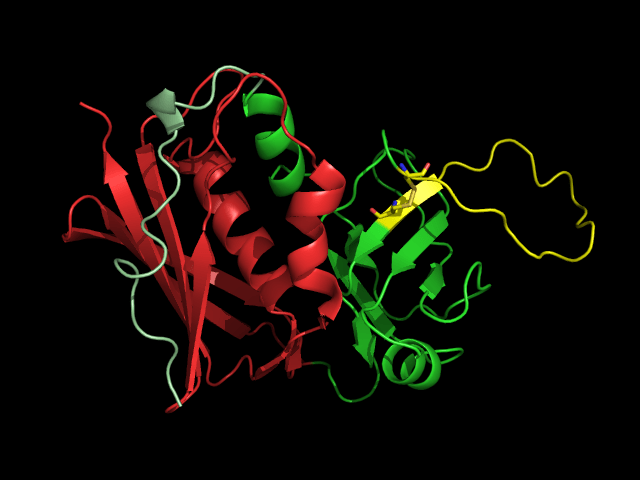
New research at the University of Chicago has uncovered the crystal structure of a protein found in the gut microbiome, providing new insights into a category of proteins once considered to be exclusively pathogenic. The study was published on September 28 in the Proceedings of the National Academy of Sciences.
This protein behaves like so-called “superantigens,” which are usually considered to be extremely pathogenic. “These are proteins made by pathogenic microbes that we think are there to either evade the immune response completely or to dysregulate it,” said senior author Erin Adams, PhD, the Joseph Regenstein Professor of Biochemistry and Molecular Biology. “These proteins bind to a large class of B cell receptors and just completely frizzle the immune system, leading to a massive inflammatory response.”
However, recent research in the Bendelac laboratory at Chicago identified a human commensal bacterium, Ruminococcus gnavus, that produces proteins called immunoglobulin-binding protein (Ibp) A and B, which appear to act like superantigens, displaying many of the same binding properties.
“Calling it a superantigen conjures thoughts of being pathogenic, but that’s not seen in this context,” said Adams. “It’s a function of the microbe; it produces this protein and presumably does so as a normal part of its interactions with other microbes in the gut.”
Using their expertise as structural biologists, the research team wanted to determine which molecular features these Ibp proteins shared with known superantigens. Isolating the complete Ibp proteins proved to be extremely challenging, but they were able to isolate a truncated version of the IbpA that could bind to mouse antigens, allowing the team to clarify the binding structure.
“We found that there are general domain structures that are shared, while there are other domains that are quite unique, without the same core structures,” said Adams. “So, we are seeing that there are some similarities between this Ibp protein and the classical superantigens, but there are some unique domains as well.”
The researchers say that understanding the structure of Ibp and how it relates to other known superantigens is important for clarifying the origins of these proteins, as well as what roles they might play in our microbiome.
“Does this mean that this protein allowed these microbes to colonize our guts without taking over as they co-evolved with humans? Was this co-opted from pathogenic bacteria to allow for some tolerance by our immune systems, or was it co-opted by the pathogenic bacteria?” said Adams. “We think that this likely evolved from a common ancestor, because it’s hard to reinvent domains, but we can’t absolutely rule out the possibility of convergent evolution. So, we have been having a lot of philosophical discussions about this!”
The similarities between the Ibp protein and other pathogenic superantigens have implications for how scientists and physicians treat diseases.
“This shows us that it would be hard to develop a silver bullet against superantigens,” said Adams. “Superantigens cause diseases like Toxic Shock Syndrome, so people can die from these things. It’s like nuclear power — it can be an amazing resource, but it can also be weaponized into something that can kill millions of people. This is the same kind of platform, that could be used in a lot of different ways. So, while it’s important to develop ways to combat them effectively, we also need to be aware of the potential effects on our microbiome.”
Future research in the Bendelac lab will focus on the physiological role of these superantigen-like proteins in the gut, and whether other similar proteins from other microbes exist.
Adams is eager to leverage the expertise in microbiome and intestinal health research at UChicago to help answer some of these questions. “We have experts here studying intestinal diseases, celiac disease, all kinds of things where we know the microbiome is involved. Does this protein play a role in those diseases? UChicago is really well suited to look at these kinds of questions.”
The study, “The molecular characterization of antibody binding to a superantigen-like protein from a commensal microbe,” was supported by the National Institute of General Medical Sciences (P30 GM124165) and an NIH-ORIP HEI grant (S10OD021527). Additional authors include Marta T. Borowska, Christoph Drees, Mayura Viswanathan, Sean M. Ryan, Jeffrey J. Bunker, and Albert Bendelac of UChicago; Alexander E. Yarawsky of the Cincinnati Children’s Hospital Medical Center; and Andrew B. Herr of the Cincinnati Children’s Hospital Medical Center and the University of Cincinnati.